Microencapsulation technology, How its work
 Microencapsulation is a smart way to improve the properties of bioactives in terms of structuration/ functionalization (e.g., converting oil into powder form, taste and odour masking, improving the flow properties of a powder, visual aspect and marketing), immobilization of volatile compounds, protection/stabilization against adverse environmental factors (e.g. during food processing and shelf life) and controlled or delayed release on the action site. This technology is applicable to a broad spectrum of compounds, from small active molecules (e.g. vitamins, minerals, flavours, oils) to bigger ones (e.g. biopolymers, enzymes, proteins) and even microorganisms.
Microencapsulation is a smart way to improve the properties of bioactives in terms of structuration/ functionalization (e.g., converting oil into powder form, taste and odour masking, improving the flow properties of a powder, visual aspect and marketing), immobilization of volatile compounds, protection/stabilization against adverse environmental factors (e.g. during food processing and shelf life) and controlled or delayed release on the action site. This technology is applicable to a broad spectrum of compounds, from small active molecules (e.g. vitamins, minerals, flavours, oils) to bigger ones (e.g. biopolymers, enzymes, proteins) and even microorganisms.
The processes involved, the formulations used, and the final physicochemical properties of the microparticles can be fine-tuned to fulfil the industrial requirements in terms of quality, volume, and cost.
Introduction of the Microencapsulation
Since the last world war, i.e. more than 70 years ago, we attended a revolution of which many of us underestimated the importance. The improvement of the agricultural production methods has led to an abundance of food in our occidental countries like mankind has never known before. Despite the criticisms made regarding the extensive use of agrochemicals, we can live now without wondering how to survive next winter. Our fridge is full!
The high performances of agriculture
The high performances of agriculture allow us to consider and include sustainable development today, even if it leads to reduction of the productivity. To reach this objective, encapsulation may bring some solutions such as slow fertilizer release (1), copper replacement by microencapsulated natural biocontrol agents (2), and inoculation of soil by immobilized rhizobacteria (3). Microencapsulation is also one effi cient way to insure good practice in livestock farming by supplementing feed with vitamins or antibiotic alternatives (4).
This food abundance is associated to a diversification of the foodstuffs
Some products, considered a few decades ago as luxury, fi nd now their place in our almost daily food (salmon, duck fi llet…), while fruits and vegetables reach us from all over the world. This evolution is linked with changes in our productivity methods. Fishing has to be replaced by aquaculture, and microencapsulation is probably one of the main technologies to develop a suitable diet for the fi shes without leading to pollution or illness propagation (5). Finally, food abundance has also driven people to request more from the food they eat, over and above energy supply; for example, to provide safety/quality, ease/convenience, health, and – why not – fun/pleasure. Microencapsulation meets all of these needs.
From traditional cooking to industrial Food industry
 From a traditional and familial cooking, we have progressively moved to industrial food. The pre-cooked food, consumed as catering (fast food) or brought back home, represents a large part of our consumption. Recomposed powders, mixes, long-term storage period, and the need for innovation have fundamentally modifi ed the handling of the foodstuffs.
From a traditional and familial cooking, we have progressively moved to industrial food. The pre-cooked food, consumed as catering (fast food) or brought back home, represents a large part of our consumption. Recomposed powders, mixes, long-term storage period, and the need for innovation have fundamentally modifi ed the handling of the foodstuffs.
From an industrial point of view, it is much more simple and less costly to transport, store, and handle powders than hydrated food products. Unfortunately, dehydration has often negative effects on the texture, flavour and solubility of the rehydrated food. Consequently, it is frequently necessary to supply food powders together with their inherent aromas, vitamins, and other properties.
Microencapsulation becomes an important tool for food process
In this context and for this particular application, microencapsulation becomes a highly important tool for food process engineers. Protected during storage or processing, released at the right time and place, the encapsulated additives will provide all their potential to the food (6). Probiotics have been defi ned as ‘‘live microorganisms that, when administered in adequate amounts, confer a health benefit on the host’’ (7).
Beneficial bacteria, such as Lactobacilli and Bifidobacterium ssp., can be found worldwide in a variety of products, including traditional/ conventional food products and dietary supplements. One of the most important prerequisites for the use of probiotics is that they survive and keep their health-promoting properties throughout the production process or during technological food treatment and storage until the end of shelf life.
Microencapsulation, an optimal strategy against adverse conditions in food
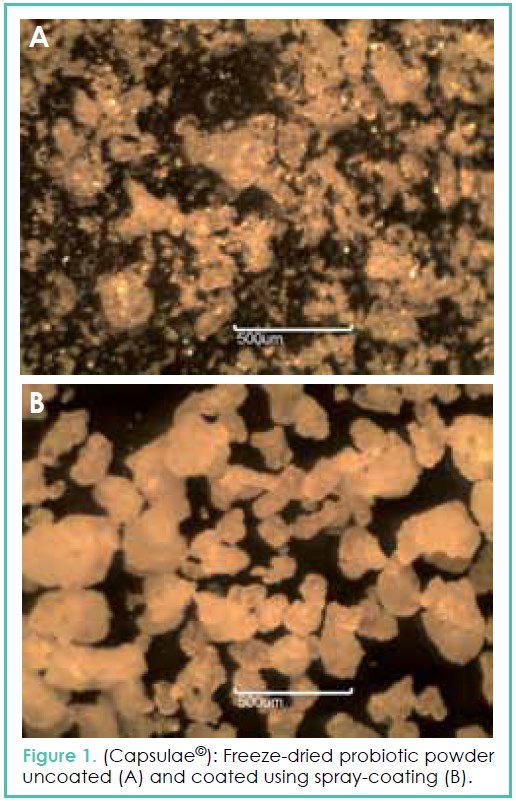
Unfortunately, the most efficient microbial strains are generally fragile cells, and therefore need to be protected. Microencapsulation of probiotics (Figure 1) can be considered an optimal strategy against adverse conditions in food and also during the gastrointestinal tract transit. It has been previously demonstrated that coating of probiotics could enhance the cell survival by a factor 100 during warm pellet extrusion (8) and by a factor 400 during storage in yogurt (9). Microencapsulation allows also the combination of probiotics with materials promoting their growth and attachment in the intestine (prebiotics).
It is therefore possible to develop optimum cocktails of probiotics and prebiotics in a single formulation called “symbiotic”. Aromas, spices and herbs constitute the core of the pleasure linked to food consumption. They also contribute to the health as nutraceuticals. During storage, freezing, and pre-cooking, they may interact with the other food ingredients, giving bad taste sometimes and losing their health potential properties. Microencapsulation allows their protection and controlled release (e.g. during cooking), offering therefore a unique approach for maintaining optimum quality and nutritive status (10).
NEW PROPERTIES AND FUNCTIONS
Functional food ingredients may be incorporated into food or may be consumed independently as pills or fine powder. This does not require a prescription from a doctor. However, consumer must be advised in order to limit its daily dose. Coating particles with a colouring material enables to differentiate functional ingredient from both a drug and a food (Figure 2).
Encapsulation can also be used as a powerful tool for innovation. A successful example is the Unistraw Delivery System (UDS), a patented straw-delivery device created in 1996 and released commercially in 2005. The UDS consists of three parts: a transparent plastic straw; patented filters inserted and welded into both ends of the straw housing; and hundreds of small beads enclosed inside the straw housing.
Flavours, vitamins and minerals, nutraceuticals and probiotics
These beads can contain (i.e. under an encapsulated form) one or several of the following compounds: flavours, vitamins and minerals, nutraceuticals and probiotics, pharmaceuticals. When the consumer sips through the straw, the liquid passes through the filters to dissolve the beads inside, delivering the flavour or functional active ingredient contained within the beads to the consumer.
Microencapsulation for food safety
Another application where encapsulation can be beneficial is food safety. Within the framework of the PROMETHEUS European FP7 Project (12), microencapsulation has recently been evaluated as a potential method to reduce consumer exposure to undesirable compounds formed during food processing without affecting food quality or microbiological safety. In particular, the potential of ingredient encapsulation to mitigate the Maillard reaction by investigating the case of sodium chloride encapsulation on the 5-hydroxymethylfurfural formation in cookies was studied (13).
The results showed that encapsulation represents a useful approach to prevent the formation of potentially harmful compounds in thermally processed foods. Finally, microencapsulation can be used as a biocatalyst immobilization system to process food in a safer and more efficient manner (14). One can cite for example the reduction of the cheese ripening time and the increase of its shelf life by processing with encapsulated enzymes (15).
DEVELOPING ENCAPSULATED FUNCTIONAL FOOD INGREDIENTS?
It exists many classifications, often complex and confusing, for the encapsulation methods. The easiest classification may be based on the equipment needed to perform the encapsulation (Figure 3).
1. Dripping consists in extruding droplets from a nozzle in gentle conditions. Droplets may be solidified by cooling or by gelation. Materials classically used as matrix include alginate, carrageenan, gellan, and pectin. Productivity may be increased by forming a liquid jet and breaking it into small droplets, multiplying nozzles or working with spinning devices.
The main advantage of this technology is the low size dispersion of the microparticles. The main drawback is the productivity.
2. Spraying is one of the oldest approaches for producing microencapsulated ingredients. The small droplets are either cooled down (molten lipid; spray-cooling) or dried (polymer solution; spray-drying). The use of spray-drying is largely developed in the food industry, as the process is very similar to the one employed to produce classical food powder (e.g. milk powder). The technology allows large productions. However, the efficacy regarding protection of the actives is not always satisfactory. Carrier materials used for spray-drying include typically gum arabic, dairy proteins, and maltodextrins.
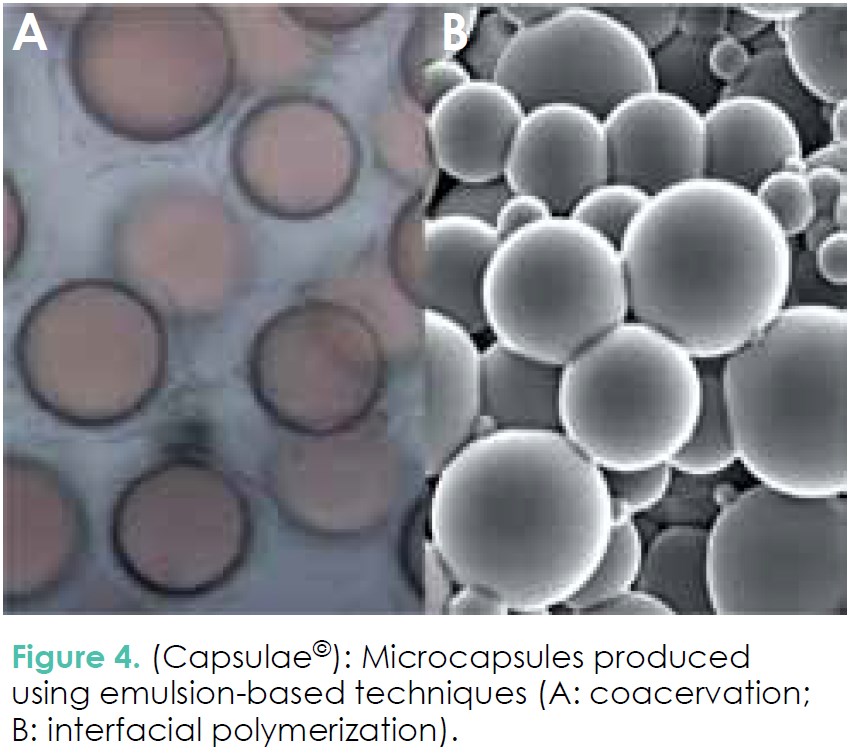
3. Dispersed emulsion droplets may be turned into microcapsules by different processes (Figure 4). In the food industry, the main method used is coacervation, which consists first in dispersing an oil phase (containing the bioactive ingredient to encapsulate) in a polymer solution, and then inducing the precipitation of the polymer(s) at the interface of the droplets (Figure 4A). If the productivity is very good, this method has an important drawback: crosslinking is necessary most of the time to obtain stable microcapsules (e.g. gelatingum arabic coacervates), which is generally done using glutaraldehyde, a non-food grade molecule prohibited in Europe for food applications. Transglutaminase, an enzymatic crosslinking agent, is often proposed as an alternative to overcome the restriction in using toxic crosslinkers.
4. Spray-coating (not to confuse with spray-drying) consists in fluidizing a powder (in a fluid bed or a pan) and spraying a coating solution on the fluidized particles. The coating is solidified by drying (polymer solution, e.g. cellulose and its derivatives) or cooling (melt system, e.g. fatty acids, waxes). This is the most promising method in terms of performances and flexibility, but process costs have to be significantly reduced.
Several constraints make the development of an encapsulation process difficult
First, encapsulation is an extra cost, which has to be minimized to be economically acceptable. This applies to the materials used to “build” the microcapsules, but also to the equipment or processing conditions. The number of food-grade materials suitable for microencapsulation is also very limited (some polysaccharides, a few lipids…). In the pharmaceutical industry, despite the strict rules to be respected for approval, many more materials are available. Consequently, the food engineer has to play finely with the coating/membrane formulation to achieve appropriate and specific properties.
CONCLUSIONS AND PERSPECTIVES
 The number of applications for microencapsulation technologies in foods, and especially in the field of functional foods and nutraceuticals, is increasing. The actual production of encapsulated food ingredients represents thousands of tons per year, and the food encapsulation technology market value is estimated to be growing at around 6 percent annually (16).
The number of applications for microencapsulation technologies in foods, and especially in the field of functional foods and nutraceuticals, is increasing. The actual production of encapsulated food ingredients represents thousands of tons per year, and the food encapsulation technology market value is estimated to be growing at around 6 percent annually (16).
Incorporation of water sensitive ingredients in high moisture foods
However, many challenges still remain. For example, incorporation of water sensitive ingredients in high moisture foods is not solved yet, because most microcapsules impermeable to water are not soft enough and will be detected by the consumers. The success of an encapsulation process is often linked to a know-how of the formulation or of the chemistry to achieve stabilisation. This is especially the case in the food industry where the number of acceptable materials is very limited, as mentioned previously.
In most cases, researchers are essentially concerned with finding the optimum formulation matching with a specific method. The “engineering” aspects are often neglected, while probably a substantial part of success to maintain the integrity of active compounds. Providing the right properties to the microcapsules and reducing the cost are directly related to the design of the equipment and the operating parameters.
The spraycoating
If the spraycoating technique is considered for example, small changes in the design of the reactor may lead to an important improvement of the process. Moving from the so-called “Würster” coating process to a spouted bed process reduces dramatically the temperature gradient inside the system (15°C), allowing an increase of the cell survival by a factor two when probiotic powders are coated (17).
Developing an encapsulated product remains a challenge
Continuous coating processes diminish the process cost by a factor 3 in comparison to equivalent batch processes (18). A control system based on pressure drop measurements has recently been developed, allowing the reduction by two of the duration of the coating process in a fluidized air bed, while avoiding agglomeration (19). Developing an encapsulated product remains a challenge, requiring a multi-disciplinary and integrated approach.
The existence of a large number of techniques as well as the multitude of parameters at stake makes difficult for a novice the choices in this field of expertise. The key for success often goes through the support and the expertise of specialized company. Furthermore, many encapsulation techniques are still at the development stage. Once fully tested and validated, these new methods will undoubtedly broaden the spectrum of possible applications for this versatile technology.
REFERENCES AND NOTES
1. Tzika M., Alexandridou S. et al., Powder Technology, 132(1), 16-24 (2003).
2. Kim I.Y., Pusey P.L. et al., Journal of Controlled Release, 161(1), 109-115 (2012).
3. Schoebitz M., Simonin H., et al., Journal of Microencapsulation, 29(6), 532-538 (2012).
4. Spanghero M., Robinson P.H. et al., Animal Feed Science and Technology, 153(1-2), 153-157 (2009).
5. Luzardo-Alvarez A., Otero-Espinar F.J. et al., Journal of Drug Delivery Science and Technology, 20(4), 277-288 (2010).
6. Zuidam N.J., Agro Food Industry Hi Tech, 21(1), 31-33 (2010).
7. FAO/WHO, Food and Agriculture Organization of the United Nations/World Health Organization, Guidelines for the Evaluation of Probiotics in Food, April 30 and May 1, 2002, London, Ontario, Canada.
8. Picot A. and Poncelet D., Proceedings of the XIII International Conference on Bioencapsulation, June 24-26, 2005, Kingston, Ontario, Canada, 31-32.
9. Picot A. and Lacroix C., International Dairy Journal, 14, 505-515 (2004).
10. Zuidam N.J. and Heinrich E., Encapsulation Technologies for Active Food Ingredients and Food Processing, Zuidam N.J. & Nedović V.A. (Eds), Springer, 127-160 (2010).
11. Nickerson M., Yan C. et al., Microencapsulation in the Food Industry – A Practical Implementation Guide, Anilkumar G., Gaonkar G. et al. (Eds), Academic Press, 485-500 (2014).
12. PROMETHEUS project (PROcess contaminants: Mitigation and Elimination Techniques for High food quality and their Evaluation Using Sensors & Simulation): https://cordis.europa.eu/project/id/265558/reporting. com/ (last checked on January 30, 2015).
13. Fiore A., Troise A.D., et al., Journal of Agricultural and Food Chemistry, 60, 10808−10814 (2012).
14. Simon J.-P. and Poncelet D., Bioencapsulation Innovations, 4(2), 14-15 (2014).
15. Kailasapathy K. and Lam S.H., International Dairy Journal, 15(6-9), 929-939 (2005).
16. Food Encapsulation Market by Technology (Microencapsulation, Macroencapsulation, Nanoencapsulation, Hybrid) by Shell Materials (Hydrocolloids, Lipids, Polymers, Melts) by Core Phase (Vitamins, Minerals, Probiotics, Colors, Flavors & Essence) – Global Trends & Forecasts to 2018: https://www.researchandmarkets.com/?gad_source=1. com/research/p3q2ch/food (last checked on January 30, 2015).
17. El mafadi S., Picot A., et al., Minerva Biotechnologica, 17, 231-235 (2006).
18. Poncelet D .and Simon J.-P., Proceedings of the XXII International Conference on Bioencapsulation, September 17-19, 2014, Bratislava, Slovakia, 84-85.
19. Prata A.S., Maudhuit A, et al., Powder Technology, 224, 168-174 (2012).
Contact-us
"*" indicates required fields



